 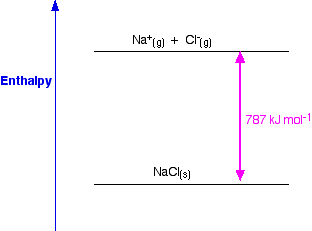
If the reaction is carried out at standard conditions, it will then be known as Standard Enthalpy Change of Reaction, ΔH θ. To start, let’s look at Enthalpy Change of Reaction, ΔH, which is defined as the heat change (heat energy absorbed or evolved) when the reaction takes place between the reagents as indicated by the stoichiometric equation for the reaction. Students need to have a good grasp of all these fundamentals before they can answer application questions on Hess’ Law, Born Haber Cycle, Energy Level Diagram, etc. If you want to master this topic, you cannot take short-cuts and expect yourself to do well. 
No wonder they are struggling with this topic. They are too “lazy” to understand and remember the key definitions of each of the different Standard Enthalpy Changes of Reactions. There are 3 sections in Chemical Energetics, namely:Īfter teaching batches of students in the last 16 years, i realised those students who are weak in this topic tends to take short-cuts. 
This topic is usually covered in term 1 or term 2 in JC1. A-Level H2 Chemistry students in junior colleges (JC) in Singapore always complain about this topic called Chemical Energetics (also known as Thermochemistry) and how difficult the questions are set in their schools. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
